Invasive disease-free survival
Improve iDFS
Help protect patients from recurrence with the Powerful Consistency of KISQALI *(1)
Add KISQALI to AI to help prevent >1 in 4 recurrences at 4 years vs AI alone. Now that is GOOD NEWS. (2)
Rate of invasive disease-free survival over 3 years. (2)
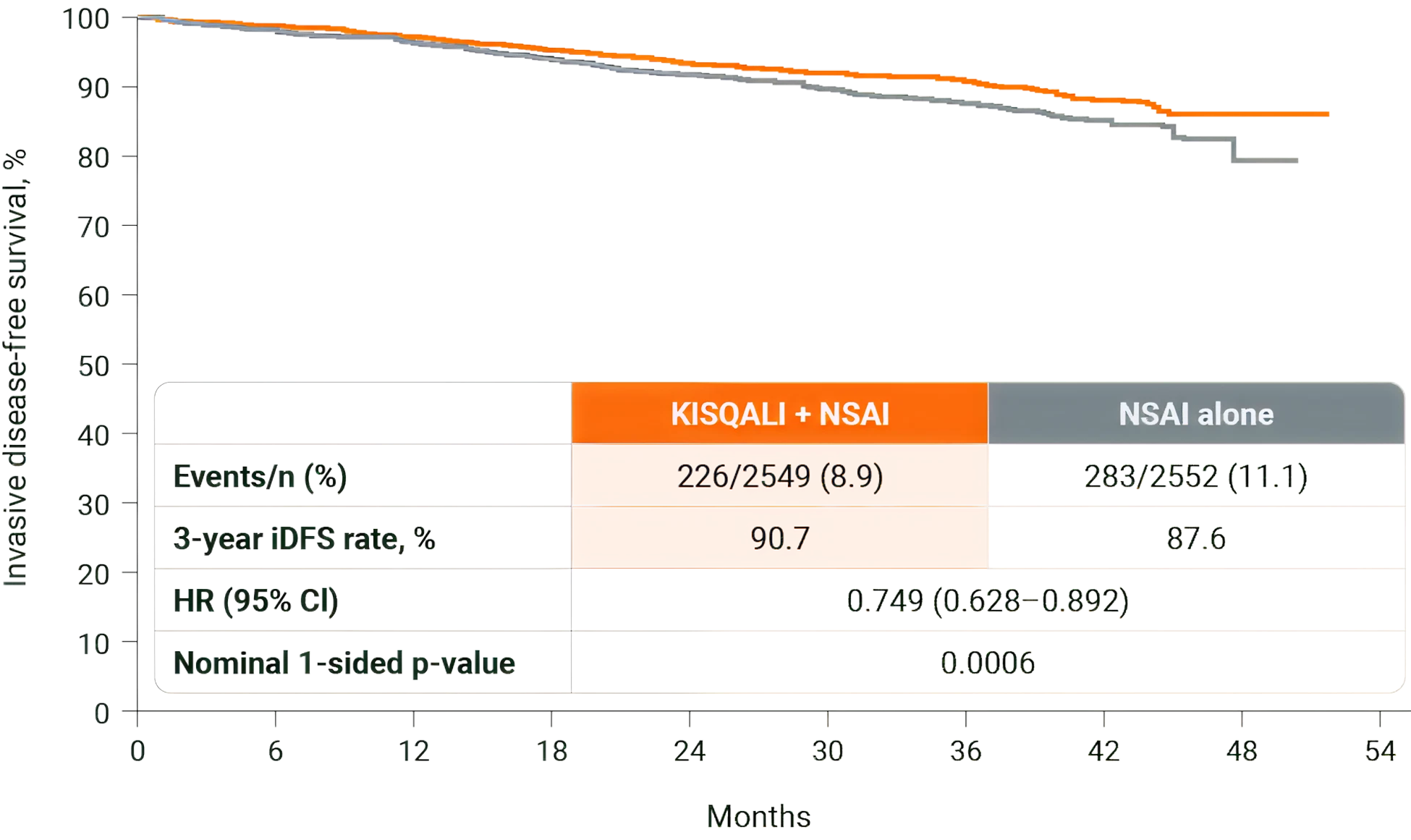
Adapted from Hortobagyi GN, et al., 2025.
Taking KISQALI for 3 years aims to reduce early and late recurrence by prolonging cell-cycle arrest and driving tumour cells into senescence/death, even after patients are off KISQALI treatment. (1,3)
Longer duration to extend time-on-target exposure and may prevent early and late recurrences. (1,3)
* vs AI in the NATALEE study and ET in the MONALEESA studies. Consistent results across a broad range of HR+/HER2– eligible patients with statistically significant improvements in iDFS (EBC), PFS and OS (ABC). (1)
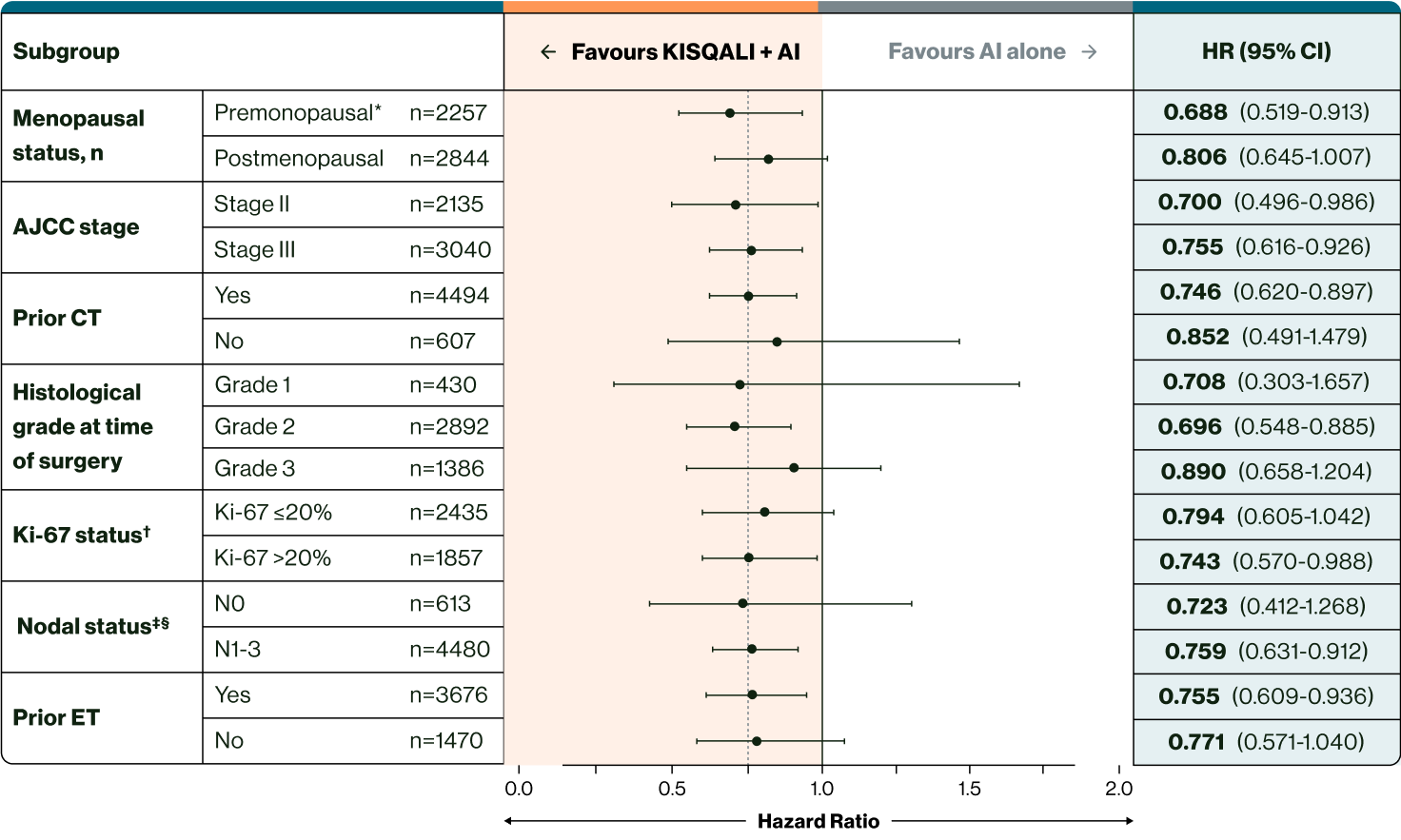
Reduced risk in all subgroups
Offer KISQALI to all N+ and high risk N0 patients who have received (neo)adjuvant ET for ≤1 year (1-4)
This reduced risk of recurrence can benefit a broad range of your eligible patients with HR+/HER2– EBC. (5)
Invasive disease-free survival risk by patient subgroup. (5)
← Slide to view more →
Adapted from Hortobagyi GN, et al., 2025.
* Included men and premenopausal women. (5)
† From archival tumour tissue. (5)
‡ Nodal status classification according to AJCC staging. (5)
§ It is important to note that for iDFS subgroup analysis, nodal status was analysed as the worst stage derived per surgical specimen or at diagnosis. (5) The values reported in the baseline characteristics were at diagnosis, so this is based on radiological evidence. Therefore, patient numbers for these subgroups may be different from those reported at diagnosis.