Dose management
Dose adjustment guidance in HR+/HER2– EBC.
Neutropenia*
Full blood counts should be performed before initiating treatment with KISQALI. (1)
After initiation of KISQALI, full blood count should be monitored every 2 weeks for the first two cycles,
at the beginning of each of the subsequent four cycles, then as clinically indicated. (1)
* Please refer to the KISQALI (ribociclib) Summary of Product Characteristics for more information on dose modification and management of neutropenia. (1)
† Grading according to Common Terminology Criteria for Adverse Events (CTCAE). (2)
Liver-related adverse events
Liver function tests should be performed before initiating treatment with KISQALI. (1)
After initiation of KISQALI, liver function tests should be performed every 2 weeks for the first two cycles, at the beginning of each of the subsequent cycles, then as clinically indicated. If grade ≥2 abnormalities are noted, more frequent monitoring is recommended. (1)
These are dose modifications for ALT and/or AST elevations from baseline without increase in total bilirubin above 2xULN. If patients develop ALT and/or AST >3x ULN along with total bilirubin >2x ULN irrespective of baseline grade, discontinue KISQALI. (1) Also, if dose reduction below 200 mg/day is required, the treatment should be permanently discontinued. (1)
* Grading according to Common Terminology Criteria for Adverse Events (CTCAE). (2)
† If baseline was normal. (2)
QT prolongation
ECG should be assessed before initiating treatment with KISQALI. (1,2)
Treatment with KISQALI should only be initiated in patients with QTcF values <450 ms. The use of KISQALI should be avoided in patients who already have or who are at significant risk of developing QT prolongation. (1)
If dose reduction <200 mg/day is required, the treatment should be permanently discontinued. Appropriate monitoring of serum electrolytes (including potassium, calcium, phosphorus and magnesium) should be performed before initiating treatment, at the beginning of the first six cycles and then as clinically indicated. Any abnormality should be corrected before initiating treatment with and during treatment with KISQALI. (1)
* In combination with Torsade de Pointes or polymorphic ventricular tachycardia or signs/symptoms of serious arrhythmia. (1)
Dose modification
KISQALI single-strength tablets make dose reduction simple and convenient. (1)
Dose reductions with KISQALI mean no need for new mid-cycle prescriptions or additional costs. (1)
Dose adjustments for AEs should be made in a stepwise order by reducing the number of tablets taken. If dose reduction below 200 mg/day is required, treatment should be permanently discontinued. (1)
Dose modification of KISQALI is recommended based on individual safety and tolerability as per the SmPC. (1)
KISQALI OS benefit is not compromised when dose reduction is needed. (2)
MONALEESA-2: OS in patients who required dose reduction. (2)
(209/334 patients) required
a KISQALI dose reduction
dose reductions were
due to adverse events
is the median time
to first dose reduction
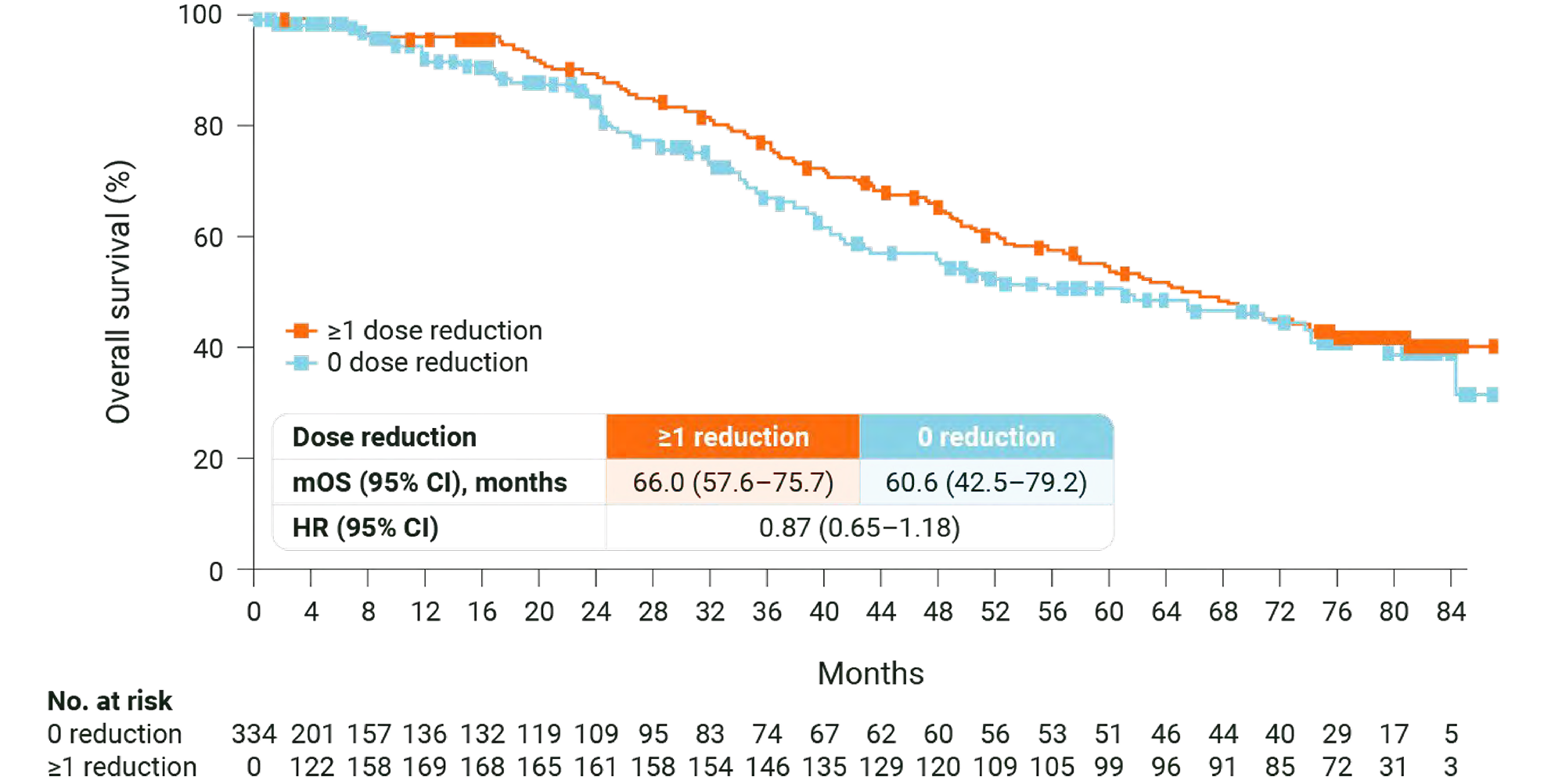
MONALEESA-2: N=668, double-blind, placebo-controlled, 1:1 randomised, multicentre, phase III trial in postmenopausal women with HR+/HER2− ABC. As 1L in advanced disease. No prior endocrine therapy for ABC and no previous systemic chemotherapy for advanced disease. KISQALI 600 mg or placebo orally once daily (3 weeks on/1 week off) + AI (letrozole 2.5 mg continuous). The primary endpoint was locally assessed PFS, and the key secondary endpoint was OS. Other secondary endpoints included the ORR (complete or partial response), the CBR (overall response plus stable disease lasting 24 weeks or more), safety, and QOL assessments. (3)


