Time to chemotherapy
Chemotherapy delay
With KISQALI you can help your patients delay chemo. (1–3)
KISQALI + ET consistently delayed the need for chemotherapy across all 3 phase III trial (1–3).
MONALEESA-2
1L postmenopausal patients
KISQALI +AI
MONALEESA-3
1L/2L postmenopausal
patients KISQALI + fulvestrant
MONALEESA-7
1L premenopausal patients
KISQALI + ET
4 years chemo-free with KISQALI + ET*(1-3)
In three key phase III trials, KISQALI + ET significantly delayed chemo vs ET alone†(1-3)
* TTC was an exploratory endpoint and was defined as the time from randomisation to the beginning of the first chemotherapy after discontinuing study treatment. (1–3)
† MONALEESA-2: HR 0.74; 95% CI: 0.61–0.91; MONALEESA-3: HR 0.70; 95% CI: 0.57–0.88; MONALEESA-7: HR 0.69; 95% CI: 0.56–0.87. (1–3)
MONALEESA trials
MONALEESA-2 study results
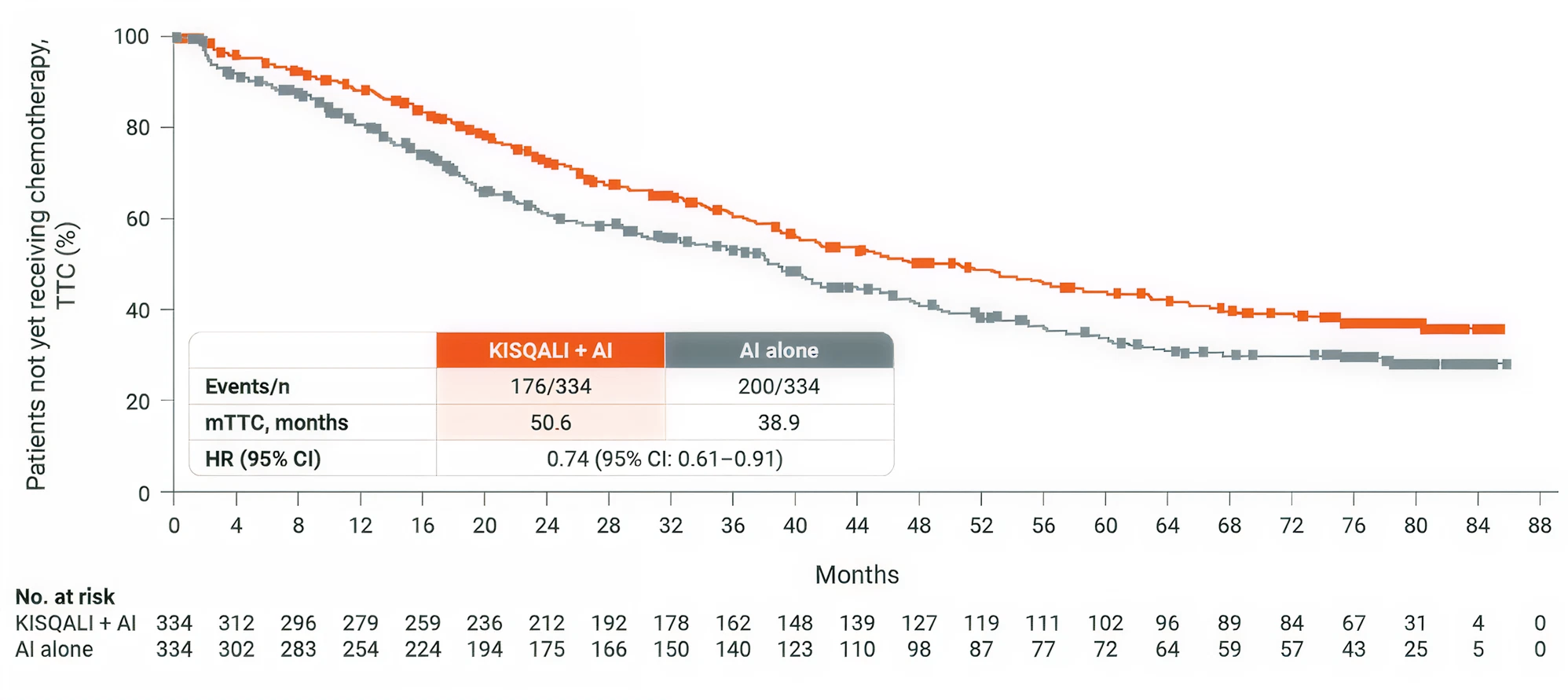
MONALEESA-2: N=668, double-blind, placebo-controlled, 1:1 randomised, multicentre, phase III trial in postmenopausal women with HR+/HER2− ABC. As 1L in advanced disease. No prior endocrine therapy for ABC and no previous systemic chemotherapy for advanced disease. KISQALI 600 mg or placebo orally once daily (3 weeks on/1 week off) + AI (letrozole 2.5 mg continuous).
The primary endpoint was locally assessed PFS, and the key secondary endpoint was OS. Other secondary endpoints included the ORR (complete or partial response), the CBR (overall response plus stable disease lasting 24 weeks or more), safety, and QOL assessments. (1,4)
MONALEESA-3 study results
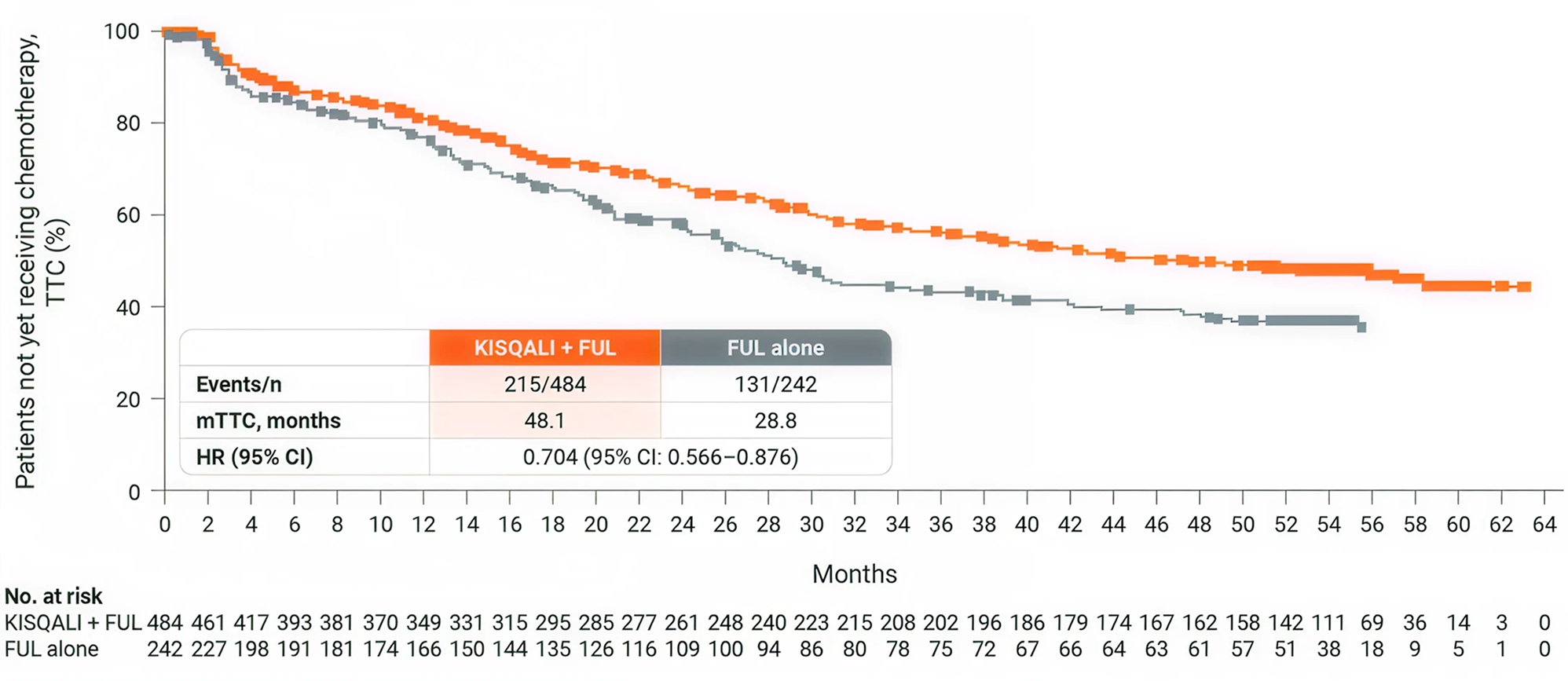
MONALEESA-3: N=726, double-blind, placebo-controlled, 2:1 randomised, phase III trial. As 1L and 2L in advanced disease plus those with early relapse in postmenopausal women with HR+/HER2– ABC. KISQALI 600 mg or placebo orally once daily (3 weeks on/1 week off) + 500 mg intramuscular fulvestrant.
The primary endpoint was locally assessed PFS. Secondary endpoints included OS, ORR, CBR, and safety and tolerability. 1L defined as: newly diagnosed (de novo) ABC patients or patients with relapse >12 months from completion of (neo)adjuvant ET with no treatment for ABC or metastatic disease. 2L was defined as: relapse on or within 12 months from completion of (neo)adjuvant endocrine therapy with no treatment for advanced or metastatic disease (early relapse); relapse >12 months from completion of (neo)adjuvant therapy with subsequent progression after one line of endocrine therapy for advanced or metastatic disease, and advanced or metastatic breast cancer at diagnosis that progressed after one line of endocrine therapy for advanced disease with no prior (neo)adjuvant treatment for early disease. (2,5)
MONALEESA-7 study results
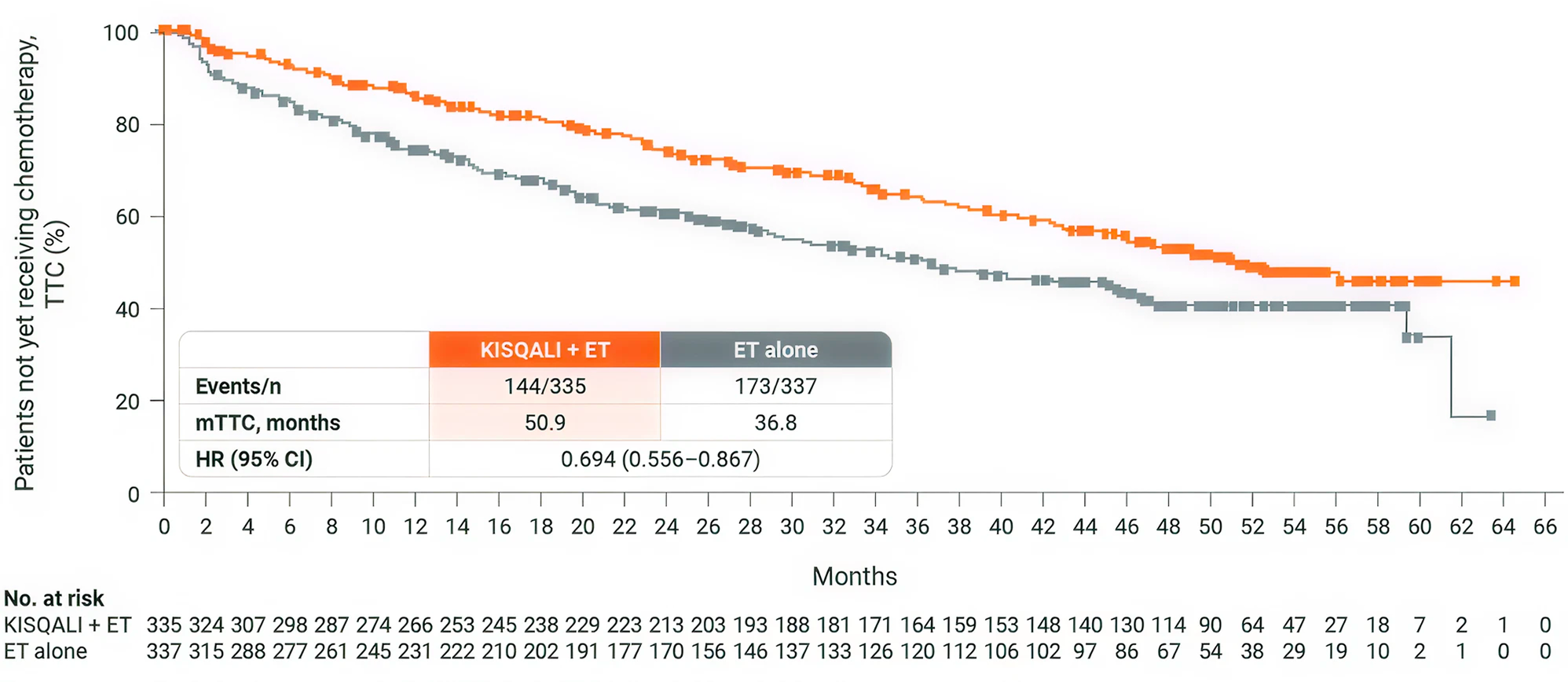
MONALEESA-7: N=672, double-blind, placebo-controlled, 1:1 randomized, phase III trial in pre- or perimenopausal women with HR+/HER2− ABC. As 1L in advanced disease and in patients who received 1 or fewer lines of chemotherapy for ABC. KISQALI 600 mg or placebo orally once daily (3 weeks on/1 week off) + AI (letrozole 2.5 mg or anastrozole 1 mg) or tamoxifen* 20 mg orally once daily continuously + LHRH agonist (goserelin 3.6 mg subcutaneously on Day 1 of every cycle). The primary endpoint was investigatorassessed PFS. The key secondary endpoint was OS, defined as the time from randomisation to death from any cause. (3)
* KISQALI should not be co-administered with tamoxifen. (6)

